Melting Points of Metals
Metals are renowned for their resilience in harsh environments, heavyweights, continuous cycling, harsh impact, corrosive conditions, and even high temperatures. High-speed equipment, combustion engines, jet engines, ignition nozzles, exhaust systems, and furnaces are frequently subjected to temperatures that can melt some metal. A number of various temperature points must be considered when choosing a metal for a high-temperature application, and one of the most important temperatures to understand is the metal melting point.
In this blog, we will go into detail about the melting points of metals and everything you need to know from importance, and understanding the highest and lowest melting points of the elements.
What is a Melting Point?
The melting point of a metal is the temperature at which it changes from a solid to a liquid state. The melting point varies depending on the specific metal.
Melting points can greatly affect your outcome depending on the project or ultimate usage. You should be aware of the melting points of the particular material utilized if you want to melt metal or expose the metal to extreme heat.
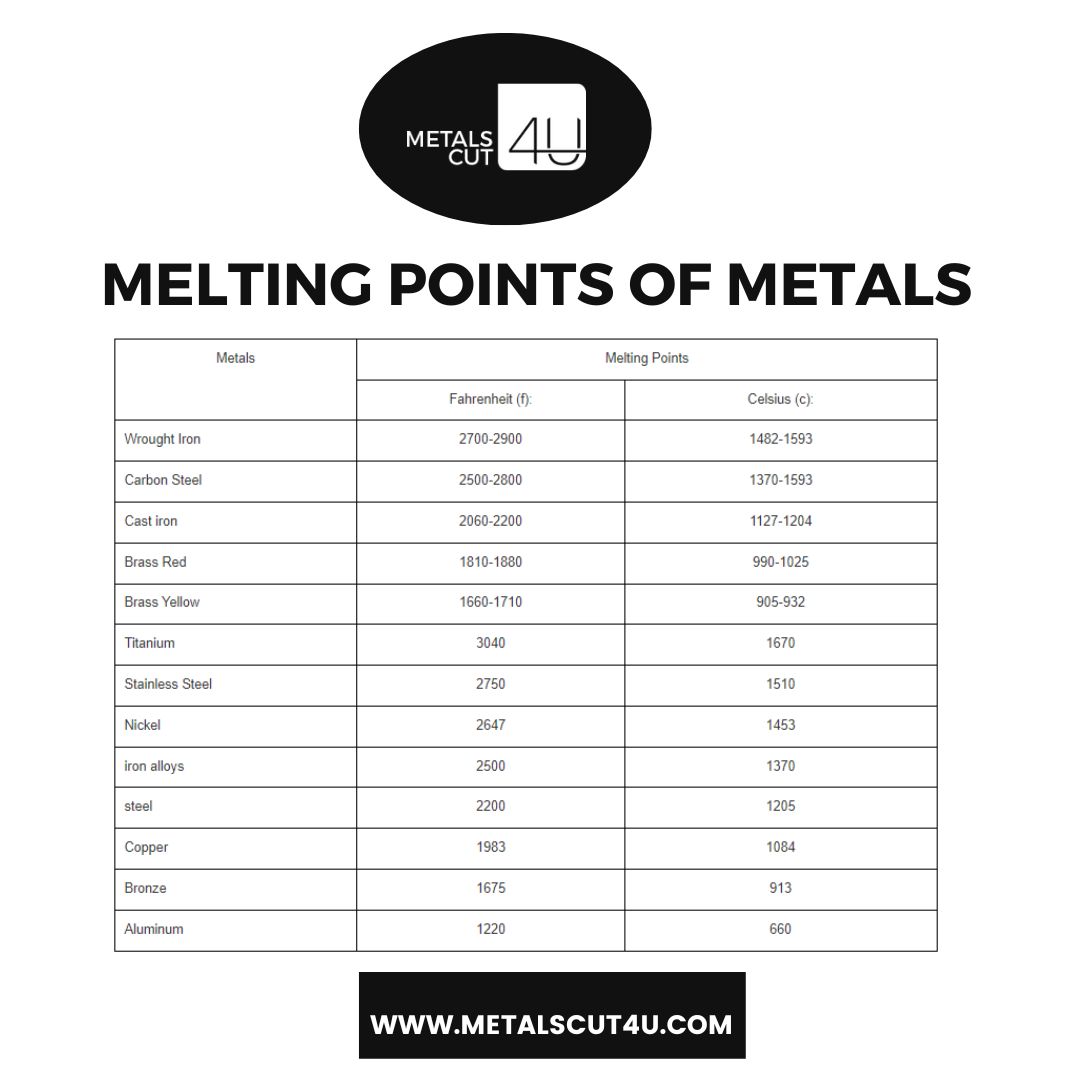
Top Metals With Their Melting Points (in Fahrenheit & Celsius )
The most popular metals used in manufacturing are shown in this melting point table in order of their melting points. These metals are more frequently used because of qualities like strength and corrosion resistance.
| Metals | Melting Points | |
| Fahrenheit (f): | Celsius (c): | |
| Wrought Iron | 2700-2900 | 1482-1593 |
| Carbon Steel | 2500-2800 | 1370-1593 |
| Cast iron | 2060-2200 | 1127-1204 |
| Brass Red | 1810-1880 | 990-1025 |
| Brass Yellow | 1660-1710 | 905-932 |
| Titanium | 3040 | 1670 |
| Stainless Steel | 2750 | 1510 |
| Nickel | 2647 | 1453 |
| iron alloys | 2500 | 1370 |
| steel | 2200 | 1205 |
| Copper | 1983 | 1084 |
| Bronze | 1675 | 913 |
| Aluminum | 1220 | 660 |
Which Metal Does Melt at The Lowest Temperature?
Mercury has the lowest melting point of most materials (and aluminum alloys for more commonly used metals). Mercury melts at a temperature of -38 °F (-39 °C), while aluminum alloys do so at a temperature of 865-1,240 °F (463-671 °C).
What is The Highest Melting Point of Any Metal?
Tungsten has the highest melting point that is on the higher end of the spectrum (and titanium for more commonly used metals). The temperature at which tungsten begins to melt is 6,150 °F (3,399 °C), while titanium begins to melt at 3,040 °F (1,670 °C).
Why Is the Melting Point of Metal Important?
The melting temperature of a metal is one of the most significant temperatures that may achieve during a metalworking process or as a result of an application. A metal can reach several other significant temperatures as well.
The component failure that might take place after a metal reaches its melting temperature is one reason why the melting temperature is so crucial. Even while metal may fail before it reaches its melting point and starts to turn into a liquid, it will no longer function as intended after it has done so. For instance, if a furnace component starts to melt and is significant enough, the furnace will no longer operate. The orifices will block if a jet engine fuel nozzle melts, making the engine inoperable. It is crucial to keep in mind that other forms of metal failure, including creep-induced fractures, may happen far before the melting temperature is reached. Therefore, study must be done beforehand on the impact of the various temperatures that metal will be exposed to.
Metals are most formable in liquid state, which is another factor that makes a metal's melting temperature crucial. For various manufacturing operations, metals are heated to their melting points. Metals must be liquid to execute smelting, fusion welding, and casting. Knowing the temperature at which the metal will melt is crucial when carrying out a manufacturing process since it enables the selection of suitable materials for the equipment being utilized. A welding gun, for instance, must be able to endure the heat generated by an electrical arc and molten metal. The dies used in casting must melt metal at a greater temperature than the metal being cast.
What Components Can Affect Metal's Melting Point?
-
External Pressure
The pressure being applied externally is the most significant factor that can increase or decrease the melting point of any metal. Because the pressure can either be caused by the normal air pressure or the pressure while conducting the reaction within a container. Therefore, melting increases the amount of the metal overall since liquids generally occupy more space than solids do.
The transformation component becomes more challenging to accomplish when the total demand is increased. It can be difficult to transition from a solid state to a liquid or gaseous state at greater external pressures since the volume needed increases rapidly. With more external pressure, more energy would be needed to cause the metal to melt.
-
Type of Bond
When determining any change in the melting point, the sort of bond your metal shares is also important to consider. Therefore, metallic compounds with ionic bonding have higher melting temperatures than those with covalent bonding.
However, because the ionic bond is integrated synonymously, more energy is often needed to disperse the bond and convert it to a liquid form. Due to the loose integration of a covalent link, melting occurs more quickly and at a lower temperature.
-
Other Metals/Impurities Present
The melting point of a metal can be altered by even the tiniest amount of impurities. Because impurities generate specific imperfections in its crystalline lattice, which make it simpler to transcend the interactions between the metal molecules, it also widens the range of melting temperatures. Therefore, the melting point can significantly and noticeably alter when any other metal is present in the mixture.
How to Avoid This Change in Melting Point?
-
Use the purest metal possible:
There are various ways to purify metal, including
-
Distillation.
-
Poling.
-
Liquation.
-
Electrolysis.
-
vapor refinement
-
zoning refinement
-
Identify the type of bond
The next stage is to determine what kind of bond the metal shares if you were successful in obtaining it in the purest form possible.
If the bond is ionic, you might need to apply more energy to get the substance to melt. You might not need to do that if the bond is covalent. You may better manage any urgent energy needs by choosing the bond type in advance.
-
Maintain Constant Pressure:
During melting, there are significant changes in the energy phases. First, ensure your container is big enough to accommodate the metal's easier contraction and expansion in any form, whether liquid or gaseous.
Tools for measuring pressure can be used to make sure that neither the internal nor external pressure changes.
Which metal is the most difficult to melt?
When it comes to melting, tungsten is the toughest member of the metal family. It does so because it has a melting point that is greater than that of any metal. It’s also one of the strongest metal on earth.
Is stainless steel's melting point a changeable temperature?
Since stainless steel contains various elements, including iron and carbon, its melting point can vary. Chromium and comparable alloying materials are also transportable.
What distinguishes the boiling point from the melting point?
Metal melting points are points at which it transforms from a solid into a liquid. A metal reaches equilibrium in the liquid and solid phases when it reaches this condition. The boiling point, on the other hand, is the temperature at which the vapor pressure of a metal equals both its ambient pressure and the pressure of any gas that is present above it.
CONFIGURE IN 4 EASY STEPS
FAQ on Melting Points of Metals
1. What temperature does metal melt?
The melting point and temperature depend on the type of metal used. For instance, steel and iron alloys require 2,200–2,500 degrees Fahrenheit (1,205–1,370 degrees Celsius) to melt completely.
2. How hot does metal have to be to melt?
As mentioned above, it depends on the temperature and type of metal used. You can check the chart displayed above for further information.
3. What metals melt easily?
With the highest melting point registered at a high temperature of 6,150 °F / 3,399 °C, Tungsten is the easiest metal to melt.
4. What metal melts at 90 degrees?
Gallium (Ga) has a melting point close to 90 degree - 86°F /30°C which can easily melt on your hand.
5. Why do metals have high melting points?
Since metal has large lattice structures, breaking countless electrostatic forces becomes difficult. Hence, metals have high melting points.
Metal melting points are important in the industrial sector.
Manufacturing of lower quality might result from any error in the melting point calculation. This is a factor that you should bear in mind if you are a producer that produces different products using metal.
Looking for a dependable manufacturer who can assist with your metal requirements, from melting to designing them into various shapes? The one you ought to pick is Metalcuts4U.
You can place an order for your own custom-cut sheet metal in just 4 simple steps. We also bend and weld it into the precise form you want before sending it your way. In only a few business days, the metal fabrication process may be completed in the amount you want for an affordable price.
